Copper(I) chloride
Did you know...
SOS Children offer a complete download of this selection for schools for use on schools intranets. Child sponsorship helps children one by one http://www.sponsor-a-child.org.uk/.
| Copper(I) chloride | |
|---|---|
 |
|
 |
|
|
Copper(I) chloride |
|
|
Other names
Cuprous chloride |
|
| Identifiers | |
| CAS number | 7758-89-6 |
| RTECS number | GL6990000 |
| Properties | |
| Molecular formula | CuCl |
| Molar mass | 98.99 g/mol |
| Appearance | white powder, slightly green from oxidized impurities |
| Density | 4.140 g/cm3, solid |
| Melting point |
430 °C (703 K) |
| Boiling point |
1490 °C (1760 K), |
| Solubility in water | 0.0062 g/100 mL (20 °C) |
| Structure | |
| Crystal structure | Tetrahedral close packed ( Zinc blende structure) |
| Hazards | |
| MSDS | ScienceLab.com |
| R/S statement | R: 22, 50, 53 S: 22, 60/61 |
| Main hazards | Irritant |
| NFPA 704 | |
| Related compounds | |
| Other anions | Copper(I) bromide Copper(I) iodide |
| Other cations | Copper(II) chloride Silver(I) chloride |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
| Infobox references | |
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. This colorless solid is a versatile precursor to other copper compounds, including some of commercial significance. It occurs naturally as the rare mineral nantokite. Unlike other first-row transition metal halides, it forma a stable complexes with carbon monoxide. It crystallizes in a diamondoid motif, reflecting the tendency of copper(I) to form tetrahedral complexes.
Preparation
Copper(I) chloride is produced industrially by the direct chlorination of copper:
- 2 Cu + Cl2 → 2 CuCl
In the laboratory, copper(I) chloride can be prepared by the reduction of copper(II) salts such as CuSO4 using sulfur dioxide, sodium bisulfite (NaHSO3), sodium metabisulfite, or copper metal.
- 2 CuCl2 + H2O + SO32- → 2 CuCl + SO42- + 2 Cl-
The white solid precipitates from the solution. Upon standing in moist air, samples of CuCl become green due to the formation of copper(II) chlorides.
Chemical properties
CuCl is more affordable and less toxic than other soft Lewis acids. In addition, copper can exist in multiple redox states, including I, II, and III. This combination of properties define some of the useful features of copper(I) chloride. It is a soft Lewis acid, classified as soft according to the Hard-Soft Acid-Base concept. Thus, it tends to form stable complexes with soft Lewis bases such as triphenylphosphine:
- CuCl + P(C6H5)3 → [CuCl(P(C6H5)3)]4
Although CuCl is insoluble in water, it dissolves in aqueous solutions containing suitable donor molecules. It forms complexes with halide ions, for example forming H3O+ CuCl2- with concentrated hydrochloric acid. It also dissolves in solutions containing CN-, S2O32-, and NH3 to give complexes.
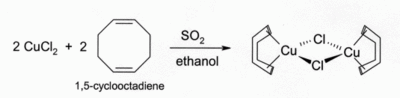
Solutions of CuCl in HCl or NH3 absorb carbon monoxide to form colourless complexes such as the chloride-bridged dimer [CuCl(CO)]2. The same hydrochloric acid solutions also react with acetylene gas to form [CuCl(C2H2)]. ammoniacal solutions of CuCl react with acetylenes to form the explosive copper(I) acetylide. Complexes of CuCl with alkenes can be prepared by reduction of CuCl2 by sulfur dioxide in the presence of the alkene in alcohol solution. Complexes with dienes such as 1,5-cyclooctadiene are particularly stable:
Although only poorly soluble in water, its aqueous solution are unstable with respect to disproportionation into Cu and CuCl2. In part for this reason samples assume a green coloration (see photograph in upper right).
Uses
The main use of copper(i) chloride is as a precursor to the fungicide copper oxychloride. For this purpose aqueous copper(I) chloride is generated by comproportionation and then air-oxidized:
- Cu + CuCl2 → 2 CuCl
- 6 CuCl + 3/2 O2 + 3 H2O → 2 Cu3Cl2(OH)4 + CuCl2
Copper(I) chloride catalyzes a variety of organic reactions, as discussed above. Its affinity for carbon monoxide in the presence of aluminium chloride is exploited in the COPureSM process.
In organic synthesis
In the Sandmeyer reaction. Treatment of an arenediazonium salt with CuCl leads to an aryl chloride, for example:
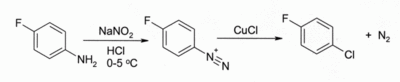 The reaction has wide scope and usually gives good yields.
The reaction has wide scope and usually gives good yields.
Early investigators observed that copper(I) halides catalyse 1,4-addition of Grignard reagents to alpha,beta-unsaturated ketones led to the development of organocuprate reagents that are widely used today in organic synthesis:
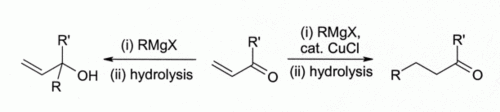 This finding led to the development of organocopper chemistry. For example, CuCl reacts with methyllithium (CH3Li) to form " Gilman reagents" such as (CH3)2CuLi, which find extensive use in organic synthesis. Grignard reagents react similarly. Although other copper(I) compounds such as copper(I) iodide are now more often used for these types of reactions, copper(I) chloride is still recommended in some case:
This finding led to the development of organocopper chemistry. For example, CuCl reacts with methyllithium (CH3Li) to form " Gilman reagents" such as (CH3)2CuLi, which find extensive use in organic synthesis. Grignard reagents react similarly. Although other copper(I) compounds such as copper(I) iodide are now more often used for these types of reactions, copper(I) chloride is still recommended in some case:
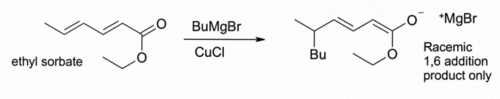 Here, Bu indicates an n- butyl group. Without CuCl, the Grignard reagent alone gives a mixture of 1,2- and 1,4-addition products (i.e., the butyl adds at the closer to the C=O).
Here, Bu indicates an n- butyl group. Without CuCl, the Grignard reagent alone gives a mixture of 1,2- and 1,4-addition products (i.e., the butyl adds at the closer to the C=O).
In polymer chemistry
Copper(I) chloride is also an intermediate formed from copper(II) chloride in the Wacker process. CuCl is used as a catalyst in Atom Transfer Radical Polymerization (ATRP).